|
12/2/2023 0 Comments Lattice energy equation ke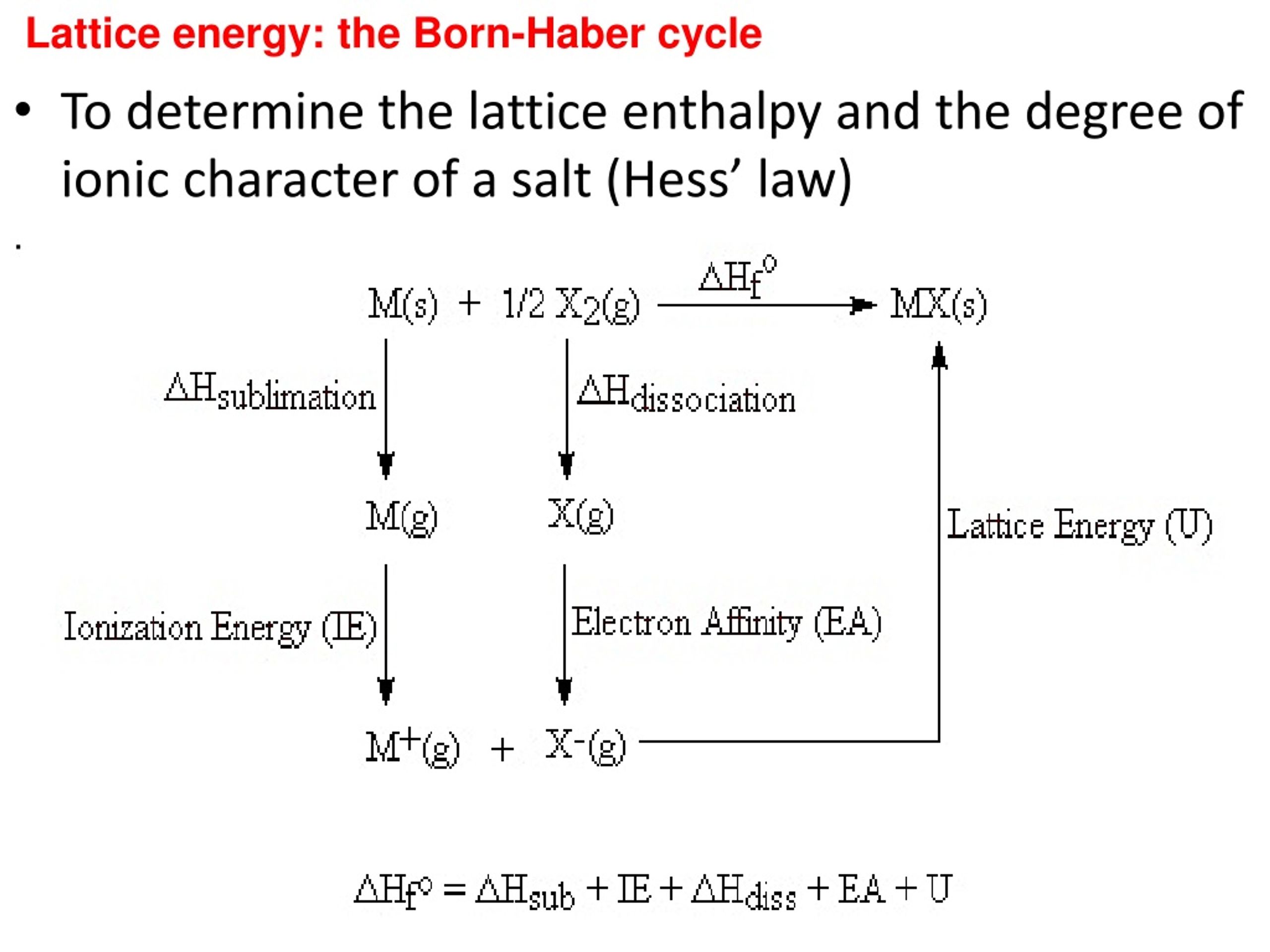
The ions are also further apart from each other in the lattice. Ionic Radius – The lattice energy becomes less exothermic as the ionic radius of the ions increases because the charge on the ions is more spread out over the ion when the ions are larger.This type of energy cannot be measured empirically, but can be calculated using electrostatics or estimated using the Born Haber cycle and its units are kJ/mol. Lattice energies are important in predicting the solubility and stability of ionic solids in water. Lattice energy also known as lattice enthalpy required to separate a mole of an ionic solid into gaseous ions or the energy that must be supplied to one mole of an ionic crystal in order to separate it into gaseous ions in a vacuum via an endothermic process. Interactive approach establishes a well-deserved academic connect between you and Master Teachers.Ahead of discussing how to calculate lattice energy, let’s define it. Sessions get recorded for you to access for quick revision later, just by a quick login to your account. Your academic progress report is shared during the Parents Teachers Meeting. Assignments, Regular Homeworks, Subjective & Objective Tests promote your regular practice of the topics. Revision notes and formula sheets are shared with you, for grasping the toughest concepts.  
WAVE platform encourages your Online engagement with the Master Teachers. We provide you year-long structured coaching classes for CBSE and ICSE Board & JEE and NEET entrance exam preparation at affordable tuition fees, with an exclusive session for clearing doubts, ensuring that neither you nor the topics remain unattended. We have grown leaps and bounds to be the best Online Tuition Website in India with immensely talented Vedantu Master Teachers, from the most reputed institutions. Vedantu LIVE Online Master Classes is an incredibly personalized tutoring platform for you, while you are staying at your home. The main factors affecting lattice energy are the charge of the constituent ions and the size of ionic radii. The lattice energy can be measured by a theoretical estimate by the Born-Lande equation or by the Born-Haber cycle which uses experimental enthalpy values. However, lattice energy measurement is difficult. It can also be defined as the energy which is needed to break the ionic compound into gaseous ions and it is an endothermic process. The lattice energy is the energy change that occurs during the formation of an ionic compound from gaseous ions which is an exothermic process. The lattice energy of the solid structure gives an approximate strength of the ionic bond. So as the atomic size of ions increases the lattice energy decreases due to the less electrostatic force between the ions. For example, considering alkali metal halides, the higher lattice energy will be for LiF and the lowest lattice energy will be for CsI. In a periodic table, as we move down a group atomic radii increase, hence lattice energy decreases down the group. So when the ions are closer, they will have higher lattice energy. 
This is because smaller ions will be closer in a lattice and thus the electrostatic attractions will be more in this case. When the ions are small, the bond between them is strong. According to the Born–Lande equation, the difference in charge numbers is the principal reason for the large difference in lattice energies. This is because BaO has charge numbers of (+2,−2) which are higher than NaCl charge numbers (+1,−1). For example, the lattice energy of NaCl is the energy change of reaction when sodium and chlorine ions react to form NaCl crystal and is equal to -786 $\dfrac$). However, it can be estimated by using the Born-Haber cycle.Īs mentioned, lattice energy is defined as the heat of formation accompanied when 1 mole of crystalline ionic solid is formed from its constituent ions which are in their gaseous state. As it is difficult to isolate gaseous ions, the experimental determination of lattice energy is difficult. Lat tice energy is the energy change accompanied when oppositely charged ions in the gas phase come together and form crystalline ionic solids. An approximate estimate of the strength of ionic bonds can be obtained by calculating l attice energy. In an ionic compound, the strength of the ionic bond between two oppositely charged ions depends mainly on the charge of ions and the distance between the ions when they are in a crystal form.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
